With this toolset you can automatically:
CLOSE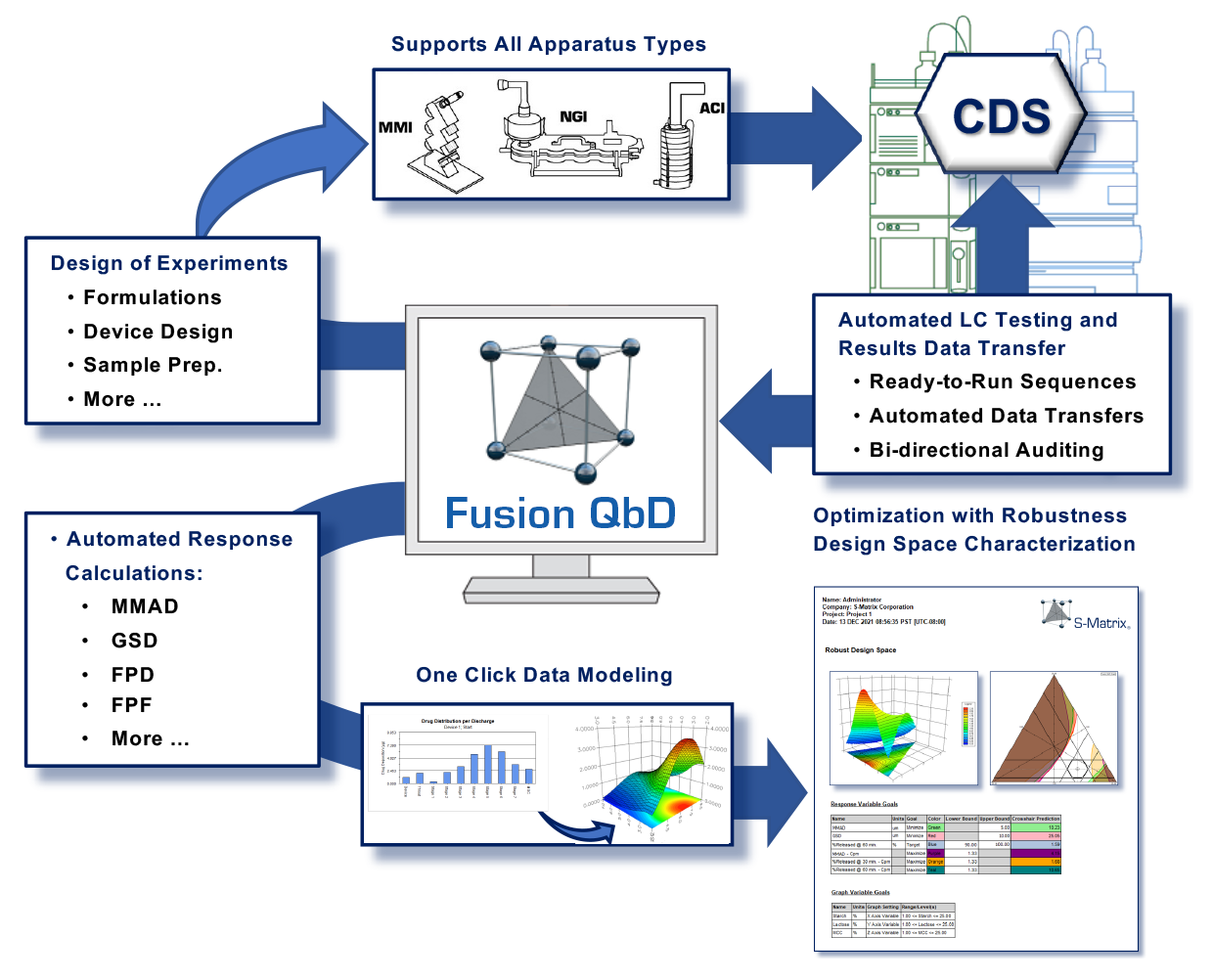
Fusion Inhaler Testing (FIT) creates Device Sampling Designs for metered-dose inhalers (MDIs) and dry powder inhalers (DPIs). It supports USP Apparatuses 1–6, Ph.Eur. Apparatuses C, D, and E, and Dose Unit Sampling Apparatuses (DUSAs). FIT also creates coordinated HPLC Testing Designs that are directly exportable to your Chromatography Data Software (CDS) — connectivity includes Agilent ChemStation, Dionex Chromeleon, and Waters Empower.

FIT Export operation automatically builds the sequence (or sample set) in the CDS, adds the appropriate standards injections, and attaches the LC Assay Method so that the sequence is ready to run on the LC in full walk-away mode.
FIT automatically imports all chromatographic results from the CDS and generates all results and graphs typically required for inhaler testing. Key features include regulatory-compliant reporting, output formats such as *.PDF and *.DOC, a full 21 CFR 11 compliance toolset, and a workflow management system.
FIT enables users to manage all of their apparatuses and product testing configurations:
FIT was developed in cooperation with several international pharmaceutical and medical device companies to address all USP and Ph.Eur analysis and reporting requirements and be consistent with current cGXP best practices.
ISO 27427 (PDF)Fusion Inhaler Testing (FIT) is the only OINDP software which supports the additional Interpolation-Regression approach to calculating MMAD and GSD specified in ISO 27427. (Page 25)
USP <1604> (PDF)USP General Chapter <1604> contains the two important elements listed below.
The change in Item 1 above impacts several important calculations based on particle-size data obtained from ACI and NGI impactors for flow rates from 30–100 L/min. The new guidance 1) provides a cutoff diameter for the pre-separator stage, and 2) excludes Stage 1 from the stages used in the calculations when the apparatus configuration does not include a pre-separator. The USP committee is currently discussing this topic, and therefore recommends not making any changes to current practice until they provide an update in 2025 — either in a revised chapter or at a USP workshop. S-Matrix will immediately incorporate any final guidance updates into our Fusion Inhaler Testing Software Platform (FIT). The next release of FIT will support the calculation approach described in Item 2 above.
To benchmark time savings using FIT versus current practice, an international pharmaceutical company repeated a standard set of testing, analysis, and reporting for an inhaled drug product testing protocol. Their estimated time savings using FIT was 40%. In addition, the automated data exchanges with the CDS virtually eliminated the possibility of transcription errors.


